Courses
The training programs are cost effective and customized based on the different levels and skill set and increase overall productivity.
At RAT CONSULTANT, through interactive training modules and state-of-the-art technology, we are able to provide an excellent platform to provide training for multiple functions on a wide range of arenas to both students and industry professionals.
We aim to support the effectiveness of the individuals by acting as a central point of contact to help Pharma students and professionals to pursue a bright future.
This intensive online training program designed by us gives you step-by-step guidance designed to support you, to remove doubt and put you in charge of configuring your own steps to a better future.
We provide with an experienced coach for support. guidance and resources to further support in understanding the concepts better.
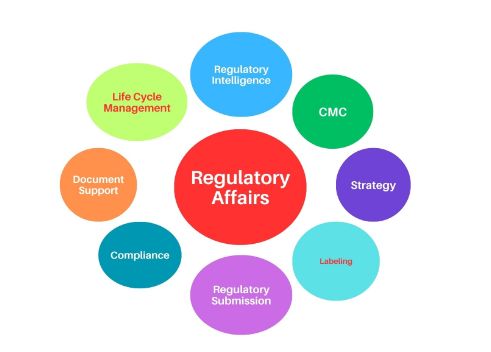
1. Foundational regulatory Knowledge
2. Effective CMC Writing and Review Skills
3. US submissions (ANDA, NDA)
4. EU submissions
5. ROW submissions
6. Dossier Harmonization
7. Post approval Submissions
8. Labelling Changes
9. ICH guidelines and adaptability in dossiers
10. Global regulatory strategies
11. Regulatory compliance and audit
12. Regulatory Intelligence (RI)
13. medical summary writing
14. Module 5 review skills